BluList - Authentag
Blulist is a newbie of Authentag to enter the field of life-sciences with their modern day tracking technology.
The Challenge
Since the new regulation was put out by FDA with a need to track all the type 2 & 3 medical devices. The client approached us to create a platform to connect FDA approved Medical device manufacturers to end users by using their tracking technology to create more users and start generating revenue with premium services.
The Solution
The project still being at a very early stage, it needed a lot of research. So, instead of coming up with final designs, we came up with three different conceptual designs that could help connect the medical manufacturers to their end users using this technology.
Here are the three different concepts
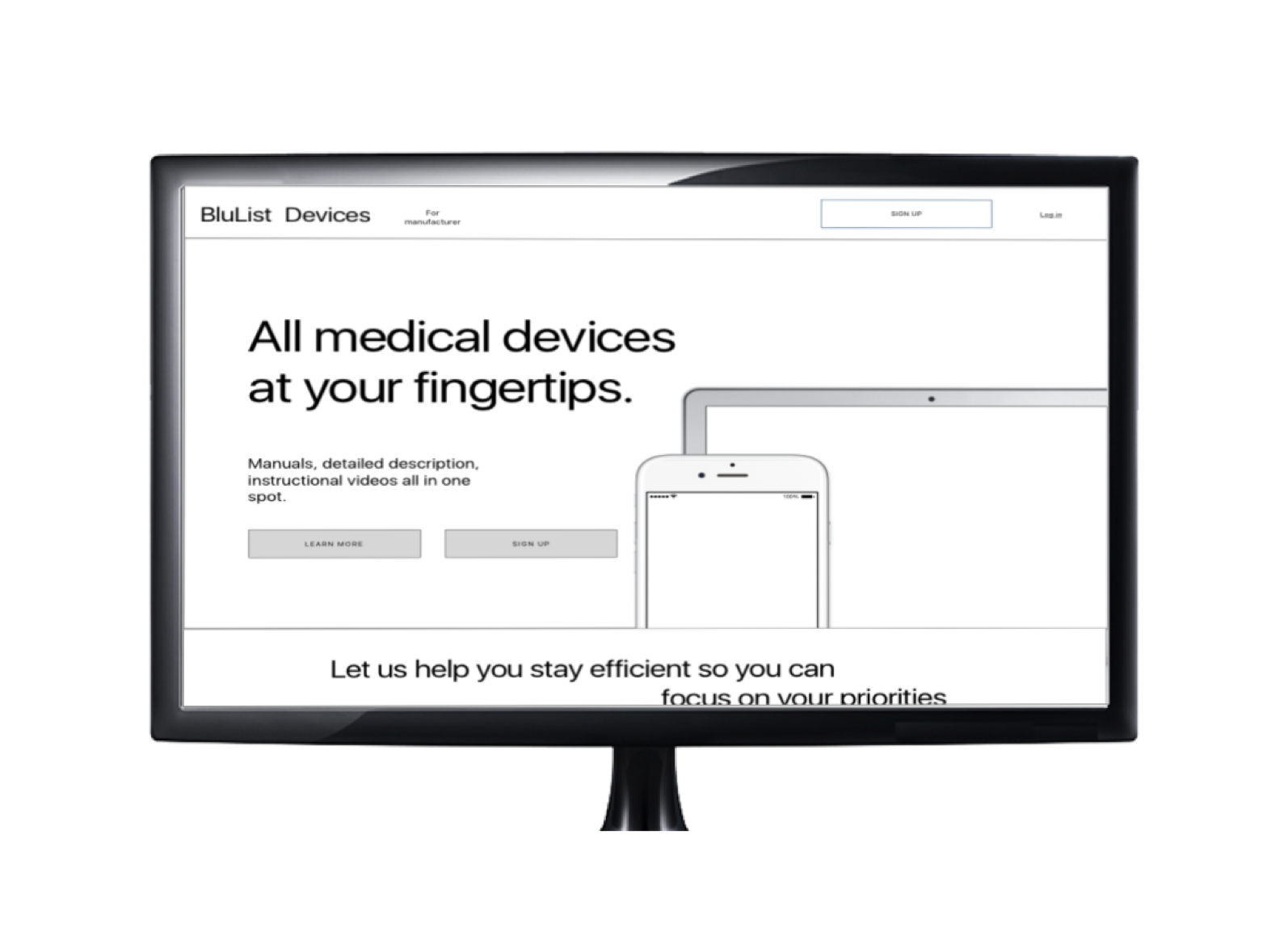
Medical professional accessing the documents.
A conceptual webpage for medical professionals to better understand the project Blulist and a search engine to enable them to search devices and get complete descriptions, manuals and instructional videos all at one place.
Medical Technician
reporting a problem.
Adding few features to already existing mobile app could ease lot of work for medical technicians like adding new logs for a device or keeping a record of all the devices etc and also could keep them in the loop with the manufacturers.
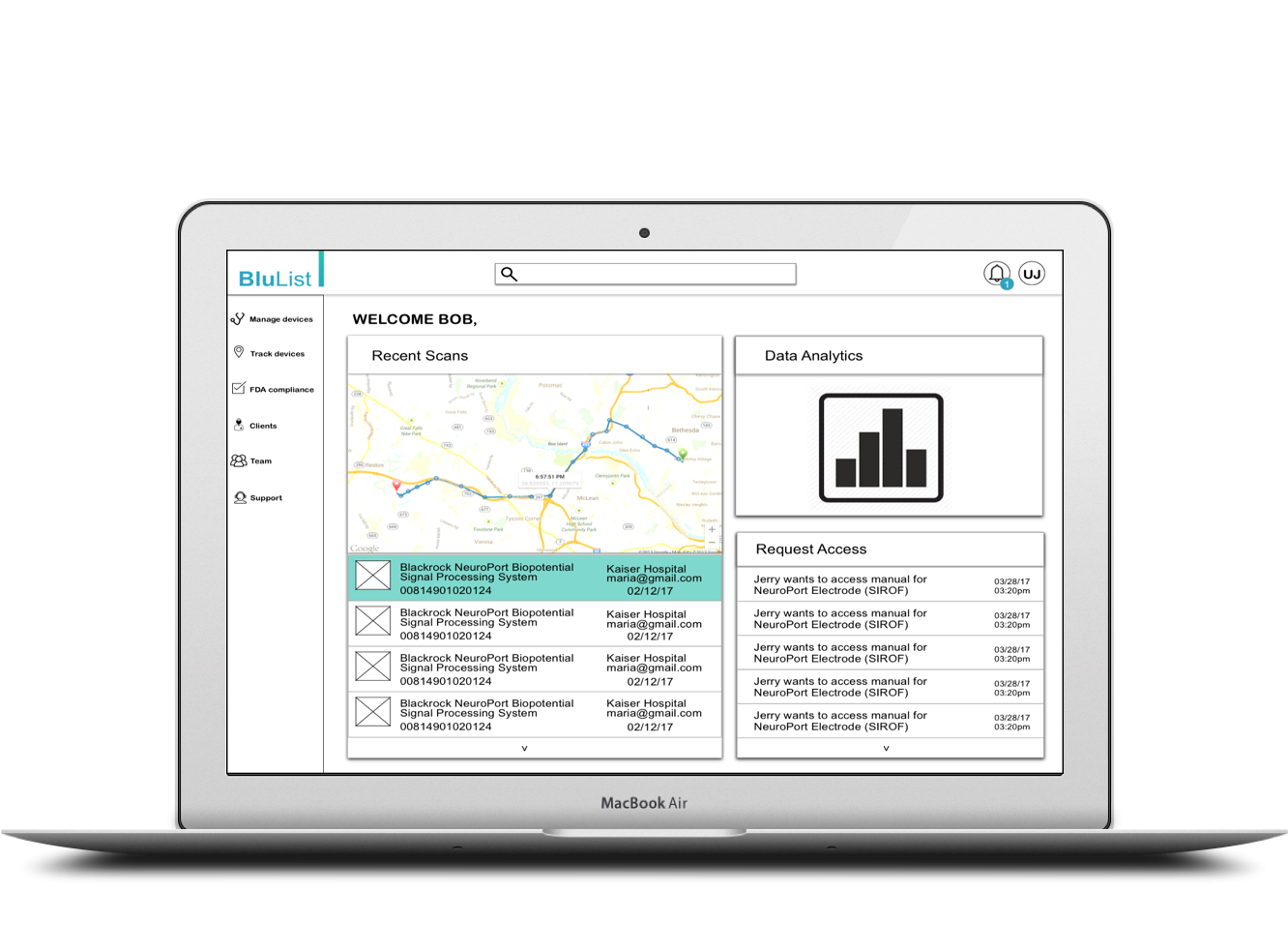
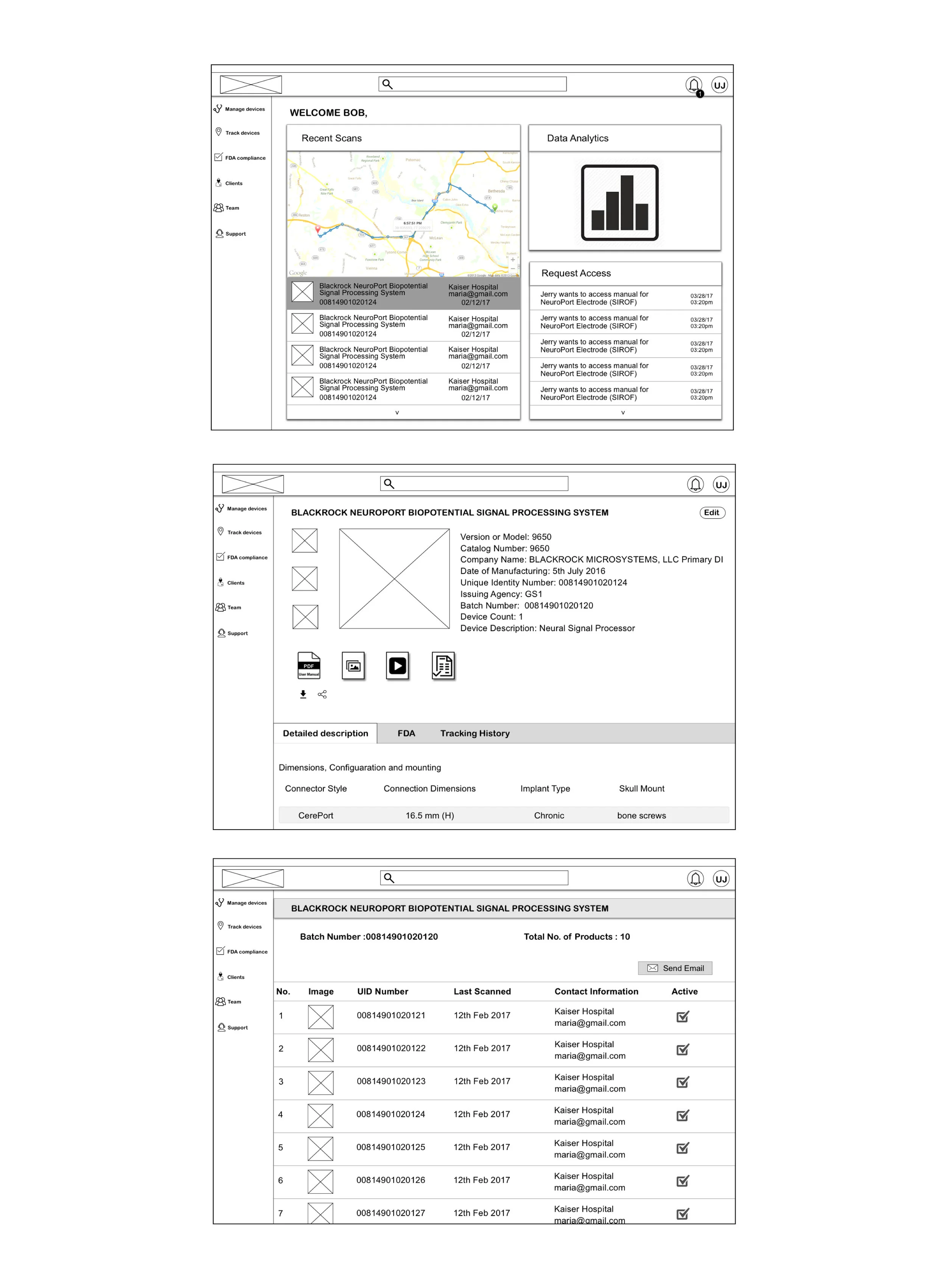
Manufacturer managing their documents
A conceptual dashboard for the manufacturers to keep all their documents in one place and to keep a track of all their devices as required by the FDA.
Design Process
Research
Getting to know the problem:
The business proposition of our client was difficult to understand, even their webpage was not clear to understand and had a lot of text. So we started our research by understanding the problem.
Market Study
Through Market study we got to know that a few other companies were tracking medical devices along with other things but had very old softwares and were not that efficient.
User Interviews
After getting a clear idea of what we wanted to know from the users, we started interviewing three main target users Medical Professionals, Medical Device manufacturers and Medical technicians. All three parties are interconnected have a deep relation.
Defining the Solution
We took three different approaches.
We need to create a medical device tracking system for:
Medical professional to make device documents available online and improve their relationship with manufacturers.
Medical technician to increase efficiency of storing device history and improve device data access.
Manufacturers to minimize costs and increase efficiency of storing device history and improve manufacturer’s compliance with FDA’s processes.
Medical professional accessing the documents
A website was the best way to create access for medical device documents to be available to medical professionals at all times on their fingertips.
System Map
2. Medical technician reporting a problem.
Instead of manually reporting every entry of the device we thought of adding a new login feature to the existing mobile app that would record all the logs of the device and at the same time send the maintainence or repair request to the manufacturer.
3. Manufacturer managing their document.
A dashboard to be provided to the manufacturers to keep all their documents such as manuals, invoices, instructional videos etc. in one place to improve their efficiency and also in case of recall. All the notifications regarding tracking a device would be provided on the dashboard. Also notifications regarding medical professionals trying to access their medical devices manuals or informational videos will be provided.